Physiology: A different cause of heart failure requires a different approach to treatment
Physiological differences affect heart failure (HF) treatment response. Studies suggest that HF has a different etiology in African Americans than in other ethnicities. While the majority of HF in white patients is caused by coronary artery disease, HF in African Americans is more likely to be caused by hypertension.1,2 Even after adjusting for risk factors, such as poor hypertension control, however, African Americans remain at higher risk for HF.1
The reason for the difference in etiology between African Americans and other ethnicities may be partially physiologic. Studies suggest that the African American population has a less active renin-angiotensin system and a lower bioavailability of nitric oxide (NO) compared with other ethnicities. This causes vasoconstriction, resulting in circulatory damage.3,4
Vasoconstriction in HF
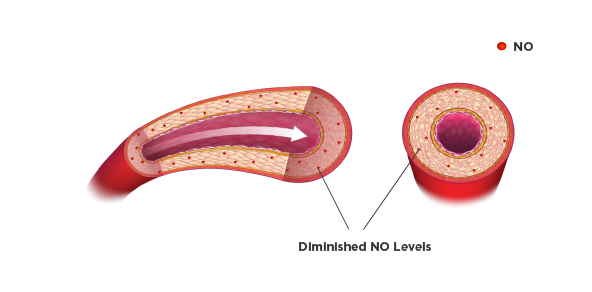
Therefore, augmentation of NO may be a useful way to slow or reverse progression of HF.4
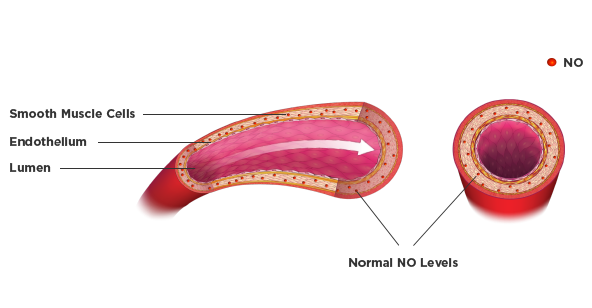
BiDil is a vasodilator, affecting both arteries and veins, that releases NO, activating guanylyl cyclase to relax vascular smooth muscle.5 It is thought that this helps to normalize NO levels, expand the veins and arteries to enhance productive blood flow, and reduce workload on the heart.Vasodilation on BiDil administration3
The mechanism of action underlying the beneficial effects of BiDil in the treatment of HF has not been fully established.5
The combination effects of BiDil
Only the fixed-dose combination of isosorbide dinitrate and hydralazine HCI—the 2 medications that compose BiDil— has been shown to provide the benefits seen in A-HeFT. Dosing the 2 medications separately has not shown bioequivalence, and thus the FDA-approved labeling for each medication component does not contain information on the treatment of heart failure.6
BiDil mechanism of action4,7
Conventional HF treatment addresses the neurohormonal excesses that contribute to the progression of disease but does not impact the bioavailability of NO. As lower levels of NO in African American patients are a potential underlying reason for the prevalence of HF, medication that augments NO may be an important adjunct to optimal HF treatment in this population.4
Heart failure therapy that addresses both pathways4,5
The mechanism of action underlying the beneficial effects of BiDil in the treatment of HF has not been established.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1-800-FDA-1088.
Pathophysiology and HFLearn how pathophysiology may play a role in how HF impacts African Americans.
INDICATIONS AND USAGE
BiDil is indicated for the treatment of heart failure as an adjunct to standard therapy in self-identified black patients to improve survival, to prolong time to hospitalization for heart failure, and to improve patient-reported functional status. There is little experience in patients with NYHA class IV heart failure. Most patients in the clinical trial supporting effectiveness (A-HeFT) received a loop diuretic, an angiotensin converting enzyme inhibitor or an angiotensin II receptor blocker, and a beta blocker, and many also received a cardiac glycoside or an aldosterone antagonist.
IMPORTANT SAFETY INFORMATION
BiDil is contraindicated in patients who are allergic to organic nitrates, or who take phosphodiesterase type 5 (PDE5) inhibitors, such as avanafil, sildenafil, tadalafil, or vardenafil, or soluble guanylate cyclase (sGC) stimulator (riociguat). Concomitant use can cause hypotension.
WARNINGS AND PRECAUTIONS
Hydralazine hydrochloride has been reported to cause a drug-induced systemic lupus erythematosus (SLE) syndrome. Symptoms and signs usually regress when hydralazine hydrochloride is discontinued.
Symptomatic hypotension, particularly with upright posture, may occur with even small doses of BiDil. Hypotension is most likely to occur in patients who have been volume or salt depleted; correct prior to initiation of BiDil. Hydralazine hydrochloride can cause tachycardia and hypotension potentially leading to myocardial ischemia and angina, particularly in patients with hypertrophic cardiomyopathy.
Hydralazine hydrochloride has been associated with peripheral neuritis, evidenced by paresthesia, numbness, and tingling, which may be related to an antipyridoxine effect. Pyridoxine should be added to BiDil therapy if such symptoms develop.
ADVERSE REACTIONS
Most common adverse reactions (> 5% more on BiDil than on placebo) were headache and dizziness.
The full Prescribing Information for BiDil is available here.
References: 1. Sharma A, Colvin-Adams M, Yancy CW. Heart failure in African Americans: disparities can be overcome. Cleve Clin J Med. 2014;81(5):301-311. 2. Yancy CW. Heart failure in African-Americans. US Cardiol. 2006;2005:2(1):196-200. 3. Yancy CW. Heart failure in African Americans: unique etiology and pharmacologic treatment responses. J Natl Med Assoc. 2003;95:1-12. 4. Taylor AL, Ziesche S, Yancy CW, et al. Combination of isosorbide dinitrate and hydralazine in blacks with heart failure. N Engl J Med. 2004;351(20):2049-2057. 5. BiDil [package insert]. Atlanta, GA: Arbor Pharmaceuticals, Inc; 2015. 6. Data on file. Arbor Pharmaceuticals, LLC. 7. Ferdinand KC, Elkayam U, Mancini D, et al. Use of isosorbide dinitrate and hydralazine in African-Americans with heart failure 9 years after the African-American Heart Failure Trial. Am J Cardiol. 2014;114:151-159.




